Advances in Auditory Interventions – A²I Lab
News
Lab Focus
The A²I Lab research program focuses on improving hearing restoration through an integrated clinical and bioengineering approach. A central goal is to address the substantial variability in outcomes observed with cochlear implants by identifying both patient-specific and device-related factors that limit performance. Clinically, this work examines indications, surgical techniques, and speech outcomes to better define the constraints of current technology. In parallel, the laboratory develops next-generation neural interfaces designed to overcome limitations of the electrode–nerve interface, a key bottleneck in cochlear implant function.
Sponsored Projects
VA-1IK2BX004910-01A2 (Brant)
For hearing loss too severe to be helped by hearing aids, cochlear implants have become the standard of care. Though they can restore the ability to understand speech for many, current electrodes have poor selectivity for neuronal excitation due to electrical current spread inherent to conventional metal electrodes. This limits fidelity leading to difficulty with background noise, talking on the phone, and music appreciation. This project aims at developing transplantable biohybrid neural interfaces specialized for the rehabilitation of hearing (“Living Electrodes”). Neuron-specific simulation permitted by this technique might allow for higher precision in stimulation of the auditory system that cannot be approached by current implant technology.
NIH-1R21DC023247-01 (Quass)
Among the one million cochlear implant (CI) patients worldwide, a substantial population persistently struggles to understand speech after implantation for often unclear reasons. Although there is substantial evidence that central plasticity contributes to a CI patient’s performance, the exact mechanisms by which CI use engages central plasticity remains unknown. We use an in-vivo approach in mice to longitudinally record the activity of cortical neuron ensembles while mice learn to use CIs, to determine if cortical plasticity is predictive of CI outcome, ultimately providing an avenue toward novel plasticity-based outcome predictors and therapies.
Lab Techniques
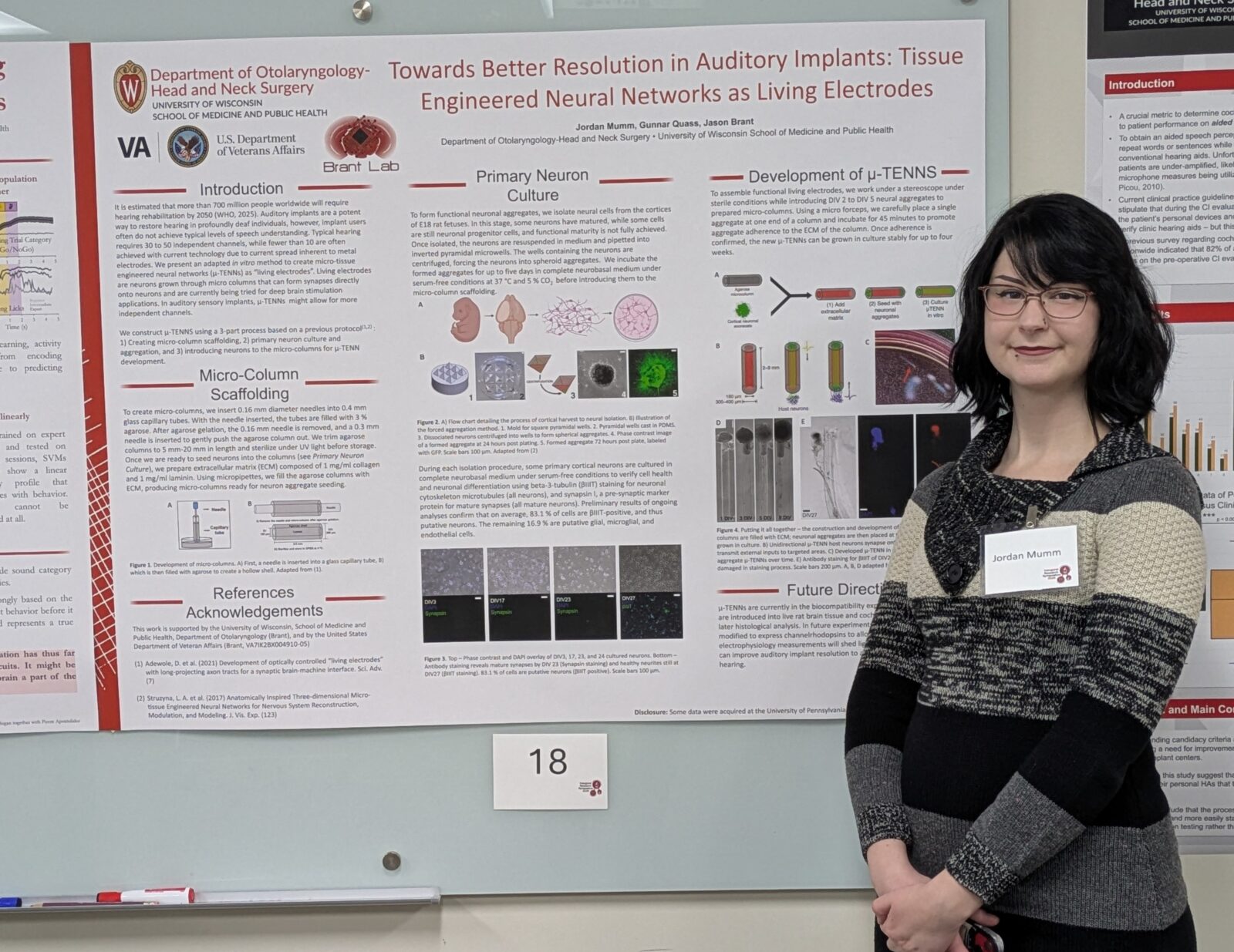
We use primary neuronal cell culture to create and maintain neuronal microspheres in-vitro. Neuronal microspheres are engineered into micro-tissue engineered neural networks (µTENNs) or “Living Electrodes” as an alternative to metal stimulation electrodes.
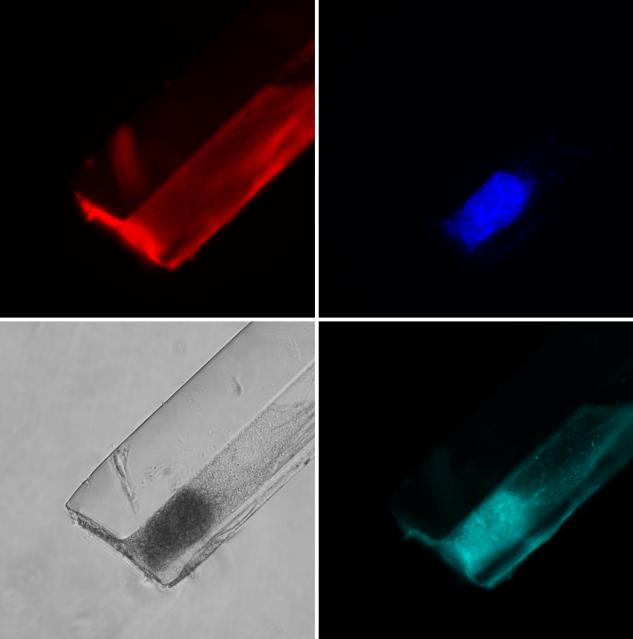
We can record brain activity of awake and anesthetized rodents listening to acoustic or electric stimuli using our 128-channel recording system in one of our two acoustically shielded recording chambers.
We explore the challenges and opportunities of artificial intelligence in hearing health care from diagnostics to cochlear implant stimulation.
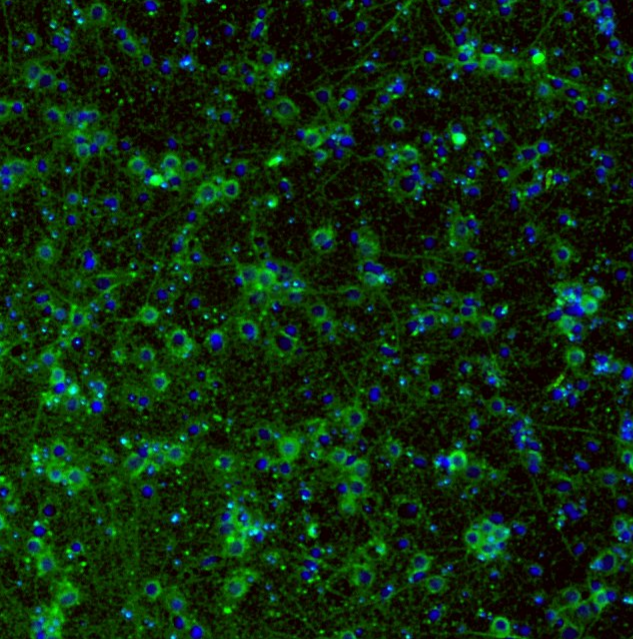
We use a variety of antibody stainings for primary 2D-cultures, neurospheres, and µ-TENNs for our experiments. Images are acquired at the WIMR imaging core.

Our ELEGOO Saturn 4 Ultra 3D printer handles all requests from simple lab experiment assistive devices to complex skull reconstructions and prototyping. We use FreeCAD to design all of our prints.

We use python or matlab to write most of our lab software ourselves, including a suite for acoustic and electric stimulation, data acquisition, and analysis programs: “Orpheus”.
Opportunities
We are looking to hire an undergraduate student in a biology or bioengineering-adjacent discipline to assist with daily lab duties and embryonic harvests. Successful candidates show interest in bioengineering and primary cell culture. Students will gain valuable experience in the inner workings of a biomedical research lab. No experience required as training will be provided. Potential duties include embryonic harvest preparation, basic cell culture, animal husbandry (rodents), immunohistochemistry. Expected commitment is 4 – 10 hours a week.
We are looking for an undergraduate student in a biology or bioengineering-adjacent discipline to evaluate primary rat cortical cultured neurons using immunocytochemistry. We require qualitative immunofluorescence images, some of which will require some protocol development, and quantitative analyses of cell counts and cell types. Flow cytometry might become a part of this project in case of early success. The project requires cell culture work, basic molecular biology and immunochemistry, microscopy, and image analysis using Fiji (ImageJ), python, or matlab. Previous experience is not necessary. Expected commitment is 4 – 10 hours a week depending on experience. This project is Bio 152 compatible.
We are looking for an undergraduate student in a biology, bioengineering, engineering, or computer science-adjacent discipline to design and test a novel fixation method for rat stereotaxic surgery. Stereotaxic surgeries are coordinate-based head surgeries and are often done using ear bars to fixate the animal. The goal of this project is to create a 3-point fixation method that leaves the ears open and instead clamps the rat at the cheekbone. The project requires 3D design in FreeCAD, 3D-resin printing, and limited interaction with rats. Previous CAD experience is valuable but not necessary. Expected commitment is 4 – 10 hours a week depending on experience. This project is Bio 152 compatible.
We are looking for an undergraduate student in a biology, bioengineering, engineering, or computer science-adjacent discipline to test a rotation correction program for rat stereotaxic surgery. Stereotaxic surgeries are coordinate-based head surgeries and can be highly susceptible to slight misalignments in animal fixation, creating major errors in targeting. The goal of this project is to test and improve correction software that translates deep brain coordinates based on measured head rotation. The project requires interaction with rats or mice, potentially including non-survival surgery, microscopy, and matlab or python programming. Previous animal handling, surgery, or programming experience is preferred but not strictly necessary. Expected commitment is 4 – 10 hours a week depending on experience. This project is Bio 152 compatible.
We have many more projects in the making that are currently not listed. Do you have a project in mind, or want to gather hands-on research experience in a future-oriented biomedical science lab? We invite motivated students of all levels to send out their CV along with a brief statement of what they are looking for.
Lab Staff

Jason A. Brant, M.D.
Associate Professor – Otolaryngology-Head and Neck Surgery
Director of Neurotology Research
jabrant@wisc.edu

Gunnar L. Quass, Ph.D.
Scientist – Otolaryngology-Head and Neck Surgery
Academic Staff Representative – District #115
gquass@wisc.edu

Jordan Mumm, B.S.
Lab Manager
Research Specialist
mumm3@wisc.edu